Tel:0086-159 8667 6306 Email:sales@alkavoda.com
Features of Alkaline Ionized Water
Views [4291] Published:2016-05-13 14:14:28
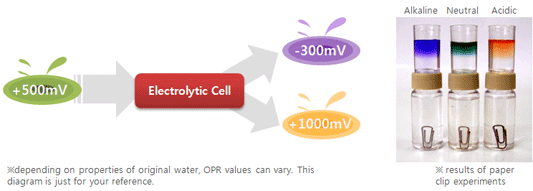
1. Has
excellent reduction potential.
ORP (Oxidation Reduction Potential): the potential for one liquid substance to reduce the oxidation of another substance, as measured in millivolts (mV). The more powerful the antioxidant, the lower the ORP level. As things oxidize, the ORP rises. example) in case of alkaline water use, anti-oxydant property can be seen; If a paper clip is put into alkaline water, rust formation will retard as compared with into tap water and acidic water. If fruits are soaked into alkaline reduced water and stored, their shelf-life will become longer. If cooked in alkaline reduced water, the foodstuffs will have longer shelf-life and will not decay rapidly.
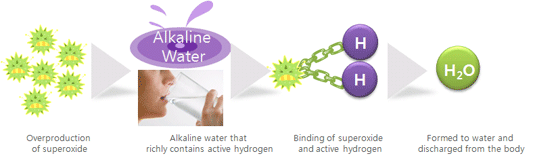
2. Active
hydrogen contained in alkaline water eliminates superoxide.
[ambivalence of superoxide] Usually: with
powerful oxidation potential it protects our body from external bacteria,
viruses, etc. If overproduced (by stress, over-ingesting acidic foods,
environmental pollution, etc.) overproduced superoxide destroys body cells and
thus can cause diseases Active hydrogen richly contained in alkaline water
eliminates superoxide in the body and helps healthy body.
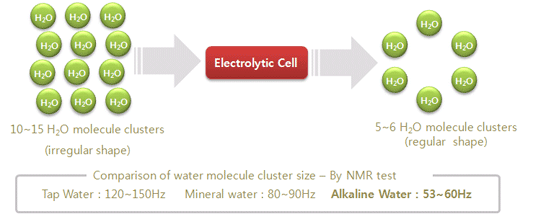
3. The
size of the cluster of water molecules is very small.
Usually 10~15 water molecules are linked to
form a ring of water, called a cluster. Clusters size of water molecules in
alkaline water becomes smaller by electrolytic process. Accordingly assimilation
into the body is far more rapid compared to other waters, and then assimilated
water rapidly discharges from the body. Besides, being small, the clusters
easily penetrate into a substance, rapidly solubilize and discharge the
resulting substance. Thus it is very good to use the alkaline water in cooking,
preparing coffee & green tea, cocktail-use ice, soaking foodstuffs for a
long time, etc.
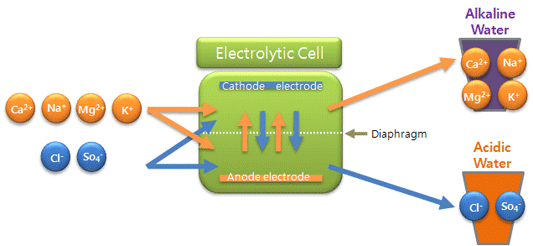
4. Mineral
content rises compared to original water.
Minerals of cationic properties contained
in water migrate into cathodic electode so that mineral content increases
compared to original water. Minerals: calcium(Ca2+), potassium(K+),
sodium(Na+), magnesium(Mg2+), etc. Cations and anions reached into each
electrolytic cell with water migrate toward the opposite direction of the
electrode, pass thru diaphragm and thus cathodic cell becomes rich in cations,
whereas anodic cell rich in anions.